Our services
We always look for ways to share and collaborate. Whether you intend to request material from our Bio-Bank, or are in need of Diagnostic Services.
The possibilities
-
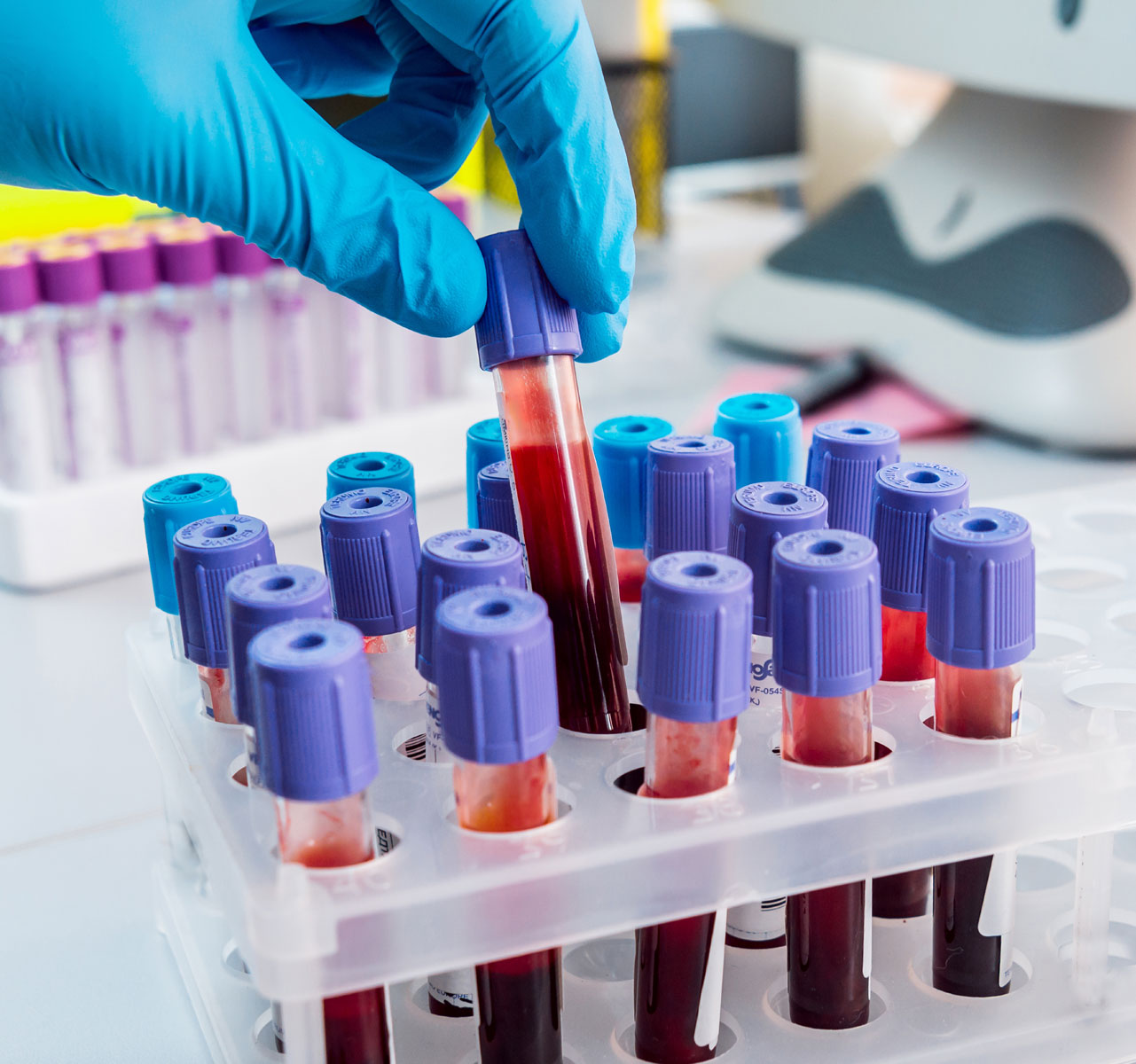
BPRC Biobank
BPRC Biobank
Biobanks are becoming a valuable and popular alternative source for exploring scientific ideas, mechanism of diseases and testing new bioactive compounds and biologicals, with the minimal possible discomfort of the animals and at low costs.
BPRC's Primate Biobank is the biggest non-human primate bank in Europe. It strives to be an international example of how to conduct the best science that meets the highest ethical standards. The biobank is based on the principals of the 3Rs: refinement, reduction and replacement in primate research.
The aim of the biobank is to provide rare and valuable primate specimens both for internal use and for external scientists who can use the material for biomedical research as well as for conservation studies.
BPRC maintains and distributes a broad range of biological material, including tissues, blood - derived products- serum, blood, DNA, RNA and B cell lines.
The transfer of the material from BPRC to the recipient can be done by:
- Signing Material Transfer Agreement (MTA) form
- Providing a CITES document for outside EU
- Providing administration costs and shipping charges by the recipient
For more details please contact: [email protected].
-
Diagnostics
Diagnostics
-
Primate Viral Diagnostics
Announcement: As per January 1th, 2026, BPRC stops offering Primate Viral Diagnostic services.
The Primate Viral Diagnostics laboratory (PVD) performs viral diagnostic research for:
- Health surveillance of the BPRC colonies
- Development and maintenance of the SPF status of the BPRC breeding colonies
- Other primate keeping facilities in Europe and other parts of the world
Below, please find an overview of the assays BPRC performs.
Serology: (serum or plasma)
Virus
- STLV (Simian T-cell lymphotropic virus)
- SIV (Simian Immunodeficiency virus)
- SRV (Simian type D retrovirus)
- SFV (Simian Foamy virus)
- α-herpesvirus (includes herpes simplex viruses 1 and 2, monkey B-virus, and others )
- γ-herpesvirus (includes SaHV2)
- VZV (Varicella Zoster virus)
- EBV (Epstein-Barr virus)
- CMV (Cytomegalovirus)
- HAV (hepatitis A virus)
- HBV (hepatitis B virus
- HCV (hepatitis C virus)
- SV40 or polyomavirus
- Influenza A and/or –B virus
- Measles virus
- Rabies virus
- Pox virus
- WNV (West Nile virus)
- DENV (Dengue virus 1-4)
- hMPV (human metapneumovirus)
- RSV (Respiratory syncytial virus)
- LCMV (Lymphocytic choriomeningitis virus)
- Various other serological tests are available on request
PCR or virus culture
- STLV PCR
- SIV PCR and culturing
- SRV type D, serotypes 1, 2 and 3 PCR
- SFV PCR and culture
- α-herpes virus PCR and culturing
- pan-herpes PCR 1 (detects α-, β-, and γ-herpesviruses)
- pan-herpes PCR 2 (detects α-, β-, and γ-herpesviruses)
- HBV PCR
- pan-polyomavirus PCR
- SV40 PCR
- Influenza A/B PCR
- Dengue virus PCR and culturing
- West Nile virus PCR and culturing
- LCMV PCR
- Various other PCRs are available on request
All PCRs can be performed in combination with sequence analysis of PCR fragment.
For more details please contact: [email protected]
-
Primate Bacterial and Parasitic Diagnostics
The Primate Bacterial and Parasitic Diagnostics laboratory of the BPRC performs bacterial and parasitic diagnostics for the health screening of the BPRC colony.
Culture
- Salmonella spp.
- Shigella spp
- Yersinia spp.
- Campylobacter spp.
- Escherichia coli O157
- Other pathogenic bacteria (upon request)
PCR
- Entamoeba histolytica
- Giardia lamblia
- Trichuris trichiura
- Helicobacter pylori
- Mycobacterium avium
Microscophy
- Helminth eggs
- Cysts
For more details please contact: [email protected]
-
Tissue analysis
The Pathology Department offers research and diagnostic pathology services for BPRC.
For more details please contact: [email protected].
-
-
Conservation Biology
Conservation Biology
Non-invasively isolated DNA from hair and stool as a source for obtaining genetic information, for the purpose of:
- Colony management of captive primates
- Genetic characterization of wild-living populations
DNA is often used in molecular biological techniques to answer diverse questions as parentage testing, colony management, forensic and immunologically related research. Hair and stool can serve as material from which DNA can be isolated, so that invasive techniques as drawing blood from an animal can be avoided. BPRC has developed techniques using different microsatellite markers, which allow to perform parentage typing and to calculate inbreeding coefficients. Other markers can be used to gain information about immunologically important genes. For example, this information is important to define the resistance or susceptibility against certain diseases and/or the influence of parasites on the genetic repertoire of an endangered primate species.
For more details please contact: [email protected].
Selected references
Determining Mhc-DRB profiles in wild populations of three congeneric true lemur species by noninvasive methods.
de Winter II, Qurkhuli T, de Groot N, de Vos-Rouweler AJM, van Hooft P, Heitkönig IMA, Prins HHT, Bontrop RE, Doxiadis GGM.
Immunogenetics. 2019 Feb;71(2):97-107. doi: 10.1007/s00251-018-1085-z. Epub 2018 Oct 15.A quick and robust MHC typing method for free-ranging and captive primate species.
de Groot N, Stanbury K, de Vos-Rouweler AJ, de Groot NG, Poirier N, Blancho G, de Luna C, Doxiadis GG, Bontrop RE.
Immunogenetics. 2017 Apr;69(4):231-240. doi: 10.1007/s00251-016-0968-0. Epub 2017 Jan 13.
-
Collaborations
Collaborations
Information on research possibilities
-
Alternatives
Each department at BPRC is involved in the development of alternatives for animal studies to fulfill the three Rs in animal research, for example the in vitro culture of malaria liver stages. Furthermore, there is a separate Alternatives department.
Alternatives department
The unit develops in vitro technologies for specific cell types or organs as an alternative for research with live animals. A second research line is the identification of new adjuvants, for example an animal friendly version of the often irreplaceable complete Freund's adjuvant by separating immune stimulatory and hazardous constituents. This unit has developed various in vitro and ex vivo models. New in vitro/ex vivo assays can be developed, depending on the questions and requests.
Contact:
Selected References
Statins amplify TLR-induced responses in microglia via inhibition of cholesterol biosynthesis.
Van Der Putten C, Kuipers HF, Zuiderwijk-Sick EA, Van Straalen L, Kondova I, Van Den Elsen PJ, Bajramovic JJ.
Glia. 2012 Jan;60(1):43-52.Alternative methods for the use of non-human primates in biomedical research.
Burm SM, Prins JB, Langermans J, Bajramovic JJ.
ALTEX. 2014;31(4):520-9.Inflammasome-induced IL-1β secretion in microglia is characterized by delayed kinetics and is only partially dependent on inflammatory caspases.
Burm SM, Zuiderwijk-Sick EA, 't Jong AE, van der Putten C, Veth J, Kondova I, Bajramovic JJ. J Neurosci. 2015 Jan 14;35(2):678-87. -
EU Sponsored Transnational Access
Installation Profile
The TRANSVAC2 consortium (website) brings together European expertise in vaccine development and testing. Biomedical researchers from industry or academia that seek access to BPRC animal models or in vitro technology, might be eligible for EU support. Please consult the website and contact us for more details.
-
Malaria
The malaria program grounds on non-human primate (NHP) models and investigates parasite-host (cell) interactions at the molecular level. In our studies we focus on NHP malaria parasites that are zoonotic, as well as on human malaria parasites.
Global response analyses
We use parasite transfection and OMIC-approaches for global response analyses as a basis for developing improved human vaccine and drug therapies. First- and second-generation vaccine candidates, based on the apical membrane antigen-1 and integrating a novel diversity-covering (DiCo) approach, were developed until the stage of clinical testing. This work included safety and immunogenicity evaluation of vaccine formulations in NHP and provided substantial expertise in recombinant protein production, vaccine formulation and immunological assay harmonization.
Ex vivo hepatocyte culture system
Next to models for in vivo and in vitro assessment of drug efficacy against blood stage malaria, we have developed a novel ex vivo hepatocyte culture system for low- to medium-throughput drug screening against (dormant) liver stages of malaria. Data acquisition is with a (confocal) high-content high-throughput imager (Operetta®) assuring data quality. This complements an in vivo drug/vaccine efficacy model in NHP to address (dormant) liver infections of NHP malaria with identical biology to human malaria.
Contact:
Selected References
Vaccination with Plasmodium knowlesi AMA1 formulated in the novel adjuvant co-vaccine HT™ protects against blood-stage challenge in rhesus macaques.
Mahdi Abdel Hamid M, Remarque EJ, van Duivenvoorde LM, van der Werff N, Walraven V, Faber BW, Kocken CH, Thomas AW.
PLoS One. 2011;6(5):e20547. doi: 10.1371/journal.pone.0020547.Humoral immune responses to a single allele PfAMA1 vaccine in healthy malaria-naïve adults.
Remarque EJ, Roestenberg M, Younis S, Walraven V, van der Werff N, Faber BW, Leroy O, Sauerwein R, Kocken CH, Thomas AW.
PLoS One. 2012;7(6):e38898. doi: 10.1371/journal.pone.0038898.Reduced CD36-dependent tissue sequestration of Plasmodium-infected erythrocytes is detrimental to malaria parasite growth in vivo.
Fonager J, Pasini EM, Braks JA, Klop O, Ramesar J, Remarque EJ, Vroegrijk IO, van Duinen SG, Thomas AW, Khan SM, Mann M, Kocken CH, Janse CJ, Franke-Fayard BM.
J Exp Med. 2012 Jan 16;209(1):93-107. doi: 10.1084/jem.20110762Transgenic fluorescent Plasmodium cynomolgi liver stages enable live imaging and purification of Malaria hypnozoite-forms.
Voorberg-van der Wel A, Zeeman AM, van Amsterdam SM, van den Berg A, Klooster EJ, Iwanaga S, Janse CJ, van Gemert GJ, Sauerwein R, Beenhakker N, Koopman G, Thomas AW, Kocken CH.
PLoS One. 2013;8(1):e54888. doi: 10.1371/journal.pone.0054888.KAI407, a potent non-8-aminoquinoline compound that kills Plasmodium cynomolgi early dormant liver stage parasites in vitro.
Zeeman AM, van Amsterdam SM, McNamara CW, Voorberg-van der Wel A, Klooster EJ, van den Berg A, Remarque EJ, Plouffe DM, van Gemert GJ, Luty A, Sauerwein R, Gagaring K, Borboa R, Chen Z, Kuhen K, Glynne RJ, Chatterjee AK, Nagle A, Roland J, Winzeler EA, Leroy D, Campo B, Diagana TT, Yeung BK, Thomas AW, Kocken CH.
Antimicrob Agents Chemother. 2014;58(3):1586-95. doi: 10.1128/AAC.01927-13.Persistence and activation of malaria hypnozoites in long-term primary hepatocyte cultures.
Dembélé L, Franetich JF, Lorthiois A, Gego A, Zeeman AM, Kocken CH, Le Grand R, Dereuddre-Bosquet N, van Gemert GJ, Sauerwein R, Vaillant JC, Hannoun L, Fuchter MJ, Diagana TT, Malmquist NA, Scherf A, Snounou G, Mazier D.
Nat Med. 2014 Mar;20(3):307-12. doi: 10.1038/nm.3461. -
Tuberculosis (TB)
The TB research program evolves around the preclinical evaluation of improved vaccine regimes by investigating aspects of vaccine safety, immunogenicity, adjuvanticity and protective efficacy in the NHP.
Measuring
Implemented and validated measures include pathology, radiography, bacteriology and clinical surrogates (hematology, clinical chemistry and metabolomics) for standardised readout of NHP TB, systemic inflammation and vaccine-mediated protection as well as for refinement of animal testing.
Identification
The research focuses on the identification of biomarkers and mechanisms of inflammation, pathogenesis and protection. Multiplex (Luminex™) assay and flow cytometry (LSR-II®, FACSaria®) protocols are established for in-depth analysis of innate and adaptive cellular immunity.
Advanced PET-CT imaging
Besides macaques (M. mulatta, M. fascicularis) we have piloted TB in common marmosets (Callithrix jacchus) as a smaller species that can be used for TB drug studies and advanced bio-imaging. The BPRC joined the Collaboration for TB Vaccine Development (CTVD, at the initiative of the Bill & Melinda Gates Foundation) as a NHP Central Service Facility and currently is implementing advanced PET-CT imaging for refined readout of NHP TB.
Contact:
Selected references
MVA.85A boosting of BCG and an attenuated, phoP deficient M. tuberculosis vaccine both show protective efficacy against tuberculosis in rhesus macaques.
Verreck FA, Vervenne RA, Kondova I, van Kralingen KW, Remarque EJ, Braskamp G, van der Werff NM, Kersbergen A, Ottenhoff TH, Heidt PJ, Gilbert SC, Gicquel B, Hill AV, Martin C, McShane H, Thomas AW.
PLoS One. 2009;4(4):e5264. doi: 10.1371/journal.pone.0005264. Epub 2009 Apr 15. Erratum in: PLoS One. 2011;6(2). doi:10.1371/annotation/e599dafd-8208-4655-a792-21cb125f7f66.Creativity in tuberculosis research and discovery.
Younga D, Verreck FA.
Tuberculosis (Edinb). 2012 Mar;92 Suppl 1:S14-6. doi: 10.1016/S1472-9792(12)70006-9. Review.Tuberculosis is associated with expansion of a motile, permissive and immunomodulatory CD16+ monocyte population via the IL-10/STAT3 axis.
Lastrucci C, Bénard A, Balboa L, Pingris K, Souriant S, Poincloux R, Al Saati T, Rasolofo V, González-Montaner P, Inwentarz S, Moranã E, Kondova I, Verreck FAW, Sasiain M, Neyrolles O, Maridonneau-Parini I, Lugo-Villarino G, and Cougoule C. (accepted for publication in Cell Research, 2015). -
Influenza
We have developed and evaluated three non-human primate species for influenza vaccine research, i.e. rhesus and cynomolgus monkeys and common marmosets. These models offer unique possibilities to study in detail the immune mechanisms and processes involved in the protection against influenza infection.
Protective effects
Our research focuses on protective effects of new vaccines, identification of correlates of protection and in depth analysis of mechanisms involved in protection. State-of-the art techniques, including imaging, in-depth analysis of cellular and humoral immune responses and pathological examination are readily available.
Contact:
Selected References
Pandemic Swine-Origin H1N1 Influenza Virus Replicates to Higher Levels and Induces More Fever and Acute Inflammatory Cytokines in Cynomolgus versus Rhesus Monkeys and Can Replicate in Common Marmosets.
Mooij P, Koopman G, Mortier D, van Heteren M, Oostermeijer H, Fagrouch Z, de Laat R, Kobinger G, Li Y, Remarque EJ, Kondova I, Verschoor EJ, Bogers WM. PLoS One. 2015 May 6;10(5):e0126132 -
HIV
BPRC has a long history in the preclinical research of new vaccine strategies for AIDS using SIV and SHIV models of the disease in rhesus monkeys. Anti-viral and gene therapy strategies to treat HIV infection have been explored in this model.
Useful model for preclinical evaluation
Due to the effectiveness of HAART therapy HIV has become a chronic infectious disease raising new scientific questions on non-AIDS related morbidities in HIV patients. Our recently developed mild SIV infection model in rhesus monkeys provides a useful model for preclinical evaluation of anti-inflammatory drugs under condition of low levels of intermittently detectable virus replication.
Contact:
Selected references
Systemic neutralizing antibodies induced by long interval mucosally primed systemically boosted immunization correlate with protection from mucosal SHIV challenge.
Bogers WM, Davis D, Baak I, Kan E, Hofman S, Sun Y, Mortier D, Lian Y, Oostermeijer H, Fagrouch Z, Dubbes R, van der Maas M, Mooij P, Koopman G, Verschoor E, Langedijk JP, Zhao J, Brocca-Cofano E, Robert-Guroff M, Srivastava I, Barnett S, Heeney JL.
Virology. 2008 Dec 20;382(2):217-25. doi: 10.1016/j.virol.2008.09.016.Acute-phase CD4+ T-cell proliferation and CD152 upregulation predict set-point virus replication in vaccinated simian-human immunodeficiency virus strain 89.6p-infected macaques.
Koopman G, Mortier D, Hofman S, Koutsoukos M, Bogers WM, Wahren B, Voss G, Heeney JL. J Gen Virol. 2009 Apr;90(Pt 4):915-26. doi: 10.1099/vir.2008.006148-0.Protection in macaques immunized with HIV-1 candidate vaccines can be predicted using the kinetics of their neutralizing antibodies.
Davis D, Koornstra W, Mortier D, Fagrouch Z, Verschoor EJ, Heeney JL, Bogers WM.
PLoS One. 2011;6(12):e28974. doi: 10.1371/journal.pone.0028974. Epub 2011 Dec 28.Immunization with apoptotic pseudovirus transduced cells induces both cellular and humoral responses: a proof of concept study in macaques.
Koopman G, Beenhakker N, Hofman S, Walther-Jallow L, Mäkitalo B, Mooij P, Anderson J, Verschoor E, Bogers WM, Heeney JL, Spetz AL.
Vaccine. 2012 Mar 28;30(15):2523-34. doi: 10.1016/j.vaccine.2012.01.082. Epub 2012 Feb 5. -
Emerging Infectious Diseases
Emerging infectious diseases pose a significant burden on global economies and public health. Often treatment of these emerging diseases is hampered by a lack of knowledge on the pathogens that cause these diseases.
Exploratory research
At BPRC, several models of emerging infectious diseases are being set up. These models, that include Dengue, West-Nile and Zika-virus, are used for exploratory research into pathogenic mechanisms and the evaluation of new vaccines and therapies against these diseases. Furthermore, BPRC has extensive knowledge on identification of novel viruses in non-human primates.
Contact:
Selected References
Characterization of novel polyomaviruses from Bornean and Sumatran orang-utans.
Groenewoud MJ, Fagrouch Z, van Gessel S, Niphuis H, Bulavaite A, Warren KS, Heeney JL, Verschoor EJ.
J Gen Virol. 2010 Mar;91(Pt 3):653-8. doi: 10.1099/vir.0.017673-0..Molecular analysis of a novel simian virus 40 (SV40) type in rhesus macaques and evidence for double infections with the classical SV40 type.
Fagrouch Z, Karremans K, Deuzing I, van Gessel S, Niphuis H, Bogers W, Verschoor EJ.
J Clin Microbiol. 2011 Apr;49(4):1280-6. doi: 10.1128/JCM.01005-10.Novel polyomaviruses in South American bats and their relationship to other members of the family Polyomaviridae.
Fagrouch Z, Sarwari R, Lavergne A, Delaval M, de Thoisy B, Lacoste V, Verschoor EJ.
J Gen Virol. 2012 Dec;93(Pt 12):2652-7. doi: 10.1099/vir.0.044149-0.Vaccine-induced protection of rhesus macaques against plasma viremia after intradermal infection with a European lineage 1 strain of West Nile virus.
Verstrepen BE, Oostermeijer H, Fagrouch Z, van Heteren M, Niphuis H, Haaksma T, Kondova I, Bogers WM, de Filette M, Sanders N, Stertman L, Magnusson S, L?rincz O, Lisziewicz J, Barzon L, Palù G, Diamond MS, Chabierski S, Ulbert S, Verschoor EJ.
PLoS One. 2014 Nov 13;9(11):e112568.Experimental infection of rhesus macaques and common marmosets with a European strain of West Nile virus.
Verstrepen BE, Fagrouch Z, van Heteren M, Buitendijk H, Haaksma T, Beenhakker N, Palù G, Richner JM, Diamond MS, Bogers WM, Barzon L, Chabierski S, Ulbert S, Kondova I, Verschoor EJ.
PLoS Negl Trop Dis. 2014 Apr 17;8(4):e2797. -
Multiple sclerosis (MS)
For the examination of the pathogenesis and treatment of MS, the primary MS model, experimental autoimmune encephalomyelitis (EAE), has been established in rhesus monkeys and in, at 8 variants, common marmosets.
Pathogenic autoimmune responses
The marmoset EAE model in particular has been successfully used in unraveling how the interaction of genetic (MHC) and environmental (infections) may elicit pathogenic autoimmune reactions leading to MS-like disease.
Biomarker indentification
In the 15 years after its introduction at BPRC the model has been used for biomarker identification in blood and urine and for the development of magnetic resonance imaging (MRI) and well-validated via a series of new immunotherapies.
Contact:
Selected references
A new primate model for multiple sclerosis in the common marmoset.
't Hart, B. A., M. van Meurs, H. P. Brok, L. Massacesi, J. Bauer, L. Boon, R. E. Bontrop, and J. D. Laman. Immunol Today. 2000. 21: 290-297.Modelling of multiple sclerosis: lessons learned in a non-human primate.
't Hart, B. A., J. D. Laman, J. Bauer, E. D. Blezer, Y. van Kooyk, and R. Q. Hintzen. The Lancet Neurology. 2004. 3: 589-597.
Non-invasive measurement of brain damage in a primate model of multiple sclerosis.
't Hart, B. A., J. T. Vogels, J. Bauer, H. P. M. Brok, and E. Blezer Trends Mol. Med. 2004. 10: 85-91.
Multiple sclerosis - a response-to-damage model.
't Hart, B. A., R. Q. Hintzen, and J. D. Laman. Trends Mol Med 2009.15: 235-244.
EAE: imperfect but useful models of multiple sclerosis.
't Hart, B. A., B. Gran, and R. Weissert. Trends Mol Med. 2011. 17: 119-125.
Improvement of preclinical animal models for autoimmune-mediated disorders via reverse translation of failed therapies.
't Hart, B. A., S. A. Jagessar, Y. S. Kap, K. G. Haanstra, I. H. Philippens, C. Serguera, J. Langermans, and M. Vierboom. Drug Discov Today. 2014. 19: 1394-1401.
EBV Infection and Multiple Sclerosis: Lessons from a Marmoset Model.
't Hart, B. A., Y. S. Kap, E. Morandi, J. D. Laman, and B. Gran.Trends Mol Med. 2016. 22: 1012-1024. -
Neurodegenerative diseases
Parkinson's disease (PD) is a progressive neurodegenerative disorder caused by degeneration of the dopamine neurons of the substantia nigra, resulting in disturbed planning and coordination of movements that seriously deteriorate quality of life.
Monitoring disease process
PD can be studied in common marmosets, based on MPTP-induced dopaminergic cell loss.
An array of computerized behavioral and neurophysiological tests has been developed for monitoring of the disease process; based on telemetry (home cage activity), touch screen (cognition tests) and EEG (sleep). The tests can be implemented in all neuropathology models.Contact:
Selected References
The MPTP marmoset model of parkinsonism: a multi-purpose non-human primate model for neurodegenerative diseases
Philippens IH, 't Hart BA, Torres G.
Drug Discov Today. 2010 Dec;15(23-24):985-90.Neurofeedback training on sensorimotor rhythm in marmoset monkeys.
Philippens IH, Vanwersch RA.
Neuroreport. 2010 Mar 31;21(5):328-32..Neuroprotective effects of riluzole in early phase Parkinson's disease on clinically relevant parameters in the marmoset MPTP model.
Verhave PS, Jongsma MJ, Van Den Berg RM, Vanwersch RA, Smit AB, Philippens IH.
Neuropharmacology. 2012 Mar;62(4):1700-7.17α-Ethynyl-androst-5-ene-3?,7β,17β-triol (HE3286) Is Neuroprotective and Reduces Motor Impairment and Neuroinflammation in a Murine MPTP Model of Parkinson's Disease.
Nicoletti F, Philippens I, Fagone P, Ahlem CN, Reading CL, Frincke JM, Auci DL.
Parkinsons Dis. 2012;2012:969418Oral treatment with the NADPH oxidase antagonist apocynin mitigates clinical and pathological features of parkinsonism in the MPTP marmoset model.
Philippens IH, Wubben JA, Finsen B, 't Hart BA.
J Neuroimmune Pharmacol. 2013 Jun;8(3):715-26. -
Genetics
Under the genetics program, BPRC monkey colonies are genetically characterized. One part of the research is focused on the major histocompatibility complex (MHC), which is a polygenic and polymorphic genomic region encoding the cell surface expressed molecules that present antigens to the immune system. The other part of the research is focussed on characterisation of the killer-cel immunoglobulin-like recepetor (KIR) expressed on natural killer (NK) cells. KIR is a ligand for MHC molecules. KIR molecules are scanning for the presence or absence of MHC on cells, for instance as a result of cancer or a viral infection, and take care of the elimination of these type of cells from the body.
Refinement and Reduction
At this level susceptibility and resistance to disease as well as the response to vaccines by the adaptive immune system is regulated. The main purpose of the genetics program is thus the definition of the most suitable animals for a specific research line, which will lead to a Refinement and Reduction of the animals used in experiments.
Contact:
Selected References
Extensive Alternative Splicing of KIR Transcripts.
Bruijnesteijn J, van der Wiel MKH, de Groot N, Otting N, de Vos-Rouweler AJM, Lardy NM, de Groot NG, Bontrop RE.
Front Immunol. 2018 Dec 4;9:2846. doi: 10.3389/fimmu.2018.02846. eCollection 2018.Human and Rhesus Macaque KIR Haplotypes Defined by Their Transcriptomes.
Bruijnesteijn J, van der Wiel MKH, Swelsen WTN, Otting N, de Vos-Rouweler AJM, Elferink D, Doxiadis GG, Claas FHJ, Lardy NM, de Groot NG, Bontrop RE.
J Immunol. 2018 Mar 1;200(5):1692-1701. doi: 10.4049/jimmunol.1701480. Epub 2018 Jan 22.Co-evolution of the MHC class I and KIR gene families in rhesus macaques: ancestry and plasticity.
de Groot NG, Blokhuis JH, Otting N, Doxiadis GG, Bontrop RE.
Immunol Rev. 2015 Sep;267(1):228-45.Differential recombination dynamics within the MHC of macaque species.
de Groot N, Doxiadis GG, Otting N, de Vos-Rouweler AJ, Bontrop RE.
Immunogenetics. 2014 Oct;66(9-10):535-44. doi: 10.1007/s00251-014-0783-4. Epub 2014 Jun 17.Haplotype diversity generated by ancient recombination-like events in the MHC of Indian rhesus macaques.
Doxiadis GG, de Groot N, Otting N, de Vos-Rouweler AJ, Bolijn MJ, Heijmans CM, de Groot NG, van der Wiel MK, Remarque EJ, Vangenot C, Nunes JM, Sanchez-Mazas A, Bontrop RE.
Immunogenetics. 2013 Aug;65(8):569-84. doi: 10.1007/s00251-013-0707-8. Epub 2013 May 29.Evolution of HLA-DRB genes.
Doxiadis GG, Hoof I, de Groot N, Bontrop RE.
Mol Biol Evol. 2012 Dec;29(12):3843-53.AIDS-protective HLA-B*27/B*57 and chimpanzee MHC class I molecules target analogous conserved areas of HIV-1/SIVcpz.
de Groot NG, Heijmans CM, Zoet YM, de Ru AH, Verreck FA, van Veelen PA, Drijfhout JW, Doxiadis GG, Remarque EJ, Doxiadis II, van Rood JJ, Koning F, Bontrop RE.
Proc Natl Acad Sci U S A. 2010 Aug 24;107(34):15175-80.

-
Please note; Any request for nonhuman primate tissues, DNA, RNA, cDNA, blood / serum samples or assays, shall be accompanied by information about the project objectives and how the requested materials or assays will be used in the project. BPRC reserves the right to decline a request for nonhuman primate tissues, DNA, RNA, cDNA, blood / serum samples or assays if, in BPRC’s opinion, such a request does not fit within the mission statement of BPRC.


