From application to experiment in animals
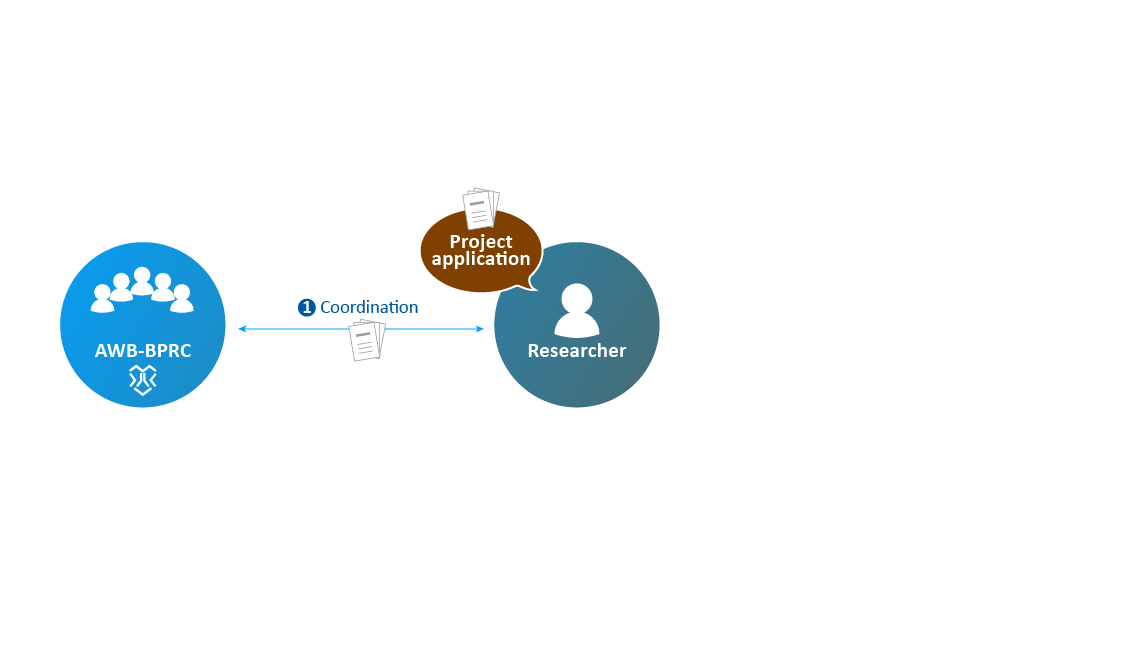
An experiment in animals is only allowed if there are no alternative methods. Prior to the actual experiment in animals, many steps must be taken.
The study should be designed in such a way that the discomfort experienced by the animals is minimised. Several authorities check if the discomfort expected outweighs the scientific interest. Here you can read how this process works.
Click on a tab for more information
-
Definitions & Abbreviations
Definitions & Abbreviations
AWB: Animal Welfare Body
The AWB-BPRC aims to ensure the optimal welfare of the laboratory animals in our institute. It is composed of our colony manager, two scientists, a veterinarian and a biotechnician.
DEC: Animal Ethics Committee
The DEC-BPRC not only includes experts on animal research, alternatives for animal research, animal protection and welfare, and applied ethics and bioethics, but also non-experts from various sections of society. To ensure the committee's independence, the chairman of the committee as well as the majority of its members are not employed by BPRC. Because the review of research proposals involving primates requires specific expertise, the DEC-BPRC is often consulted by the CCD (see below) during the reviewing process. Of course, the CCD can also decide to consult another DEC.
CCD: Central Authority for Scientific Procedures on Animals
This national authority issues a "project licence" to the licence holder.
NVWA: Netherlands Food and Consumer Product Safety Authority
This national body supervises animal welfare and the execution of animal experiments.
-
Process
Process
Use the arrows in the illustration or swipe for the next step.
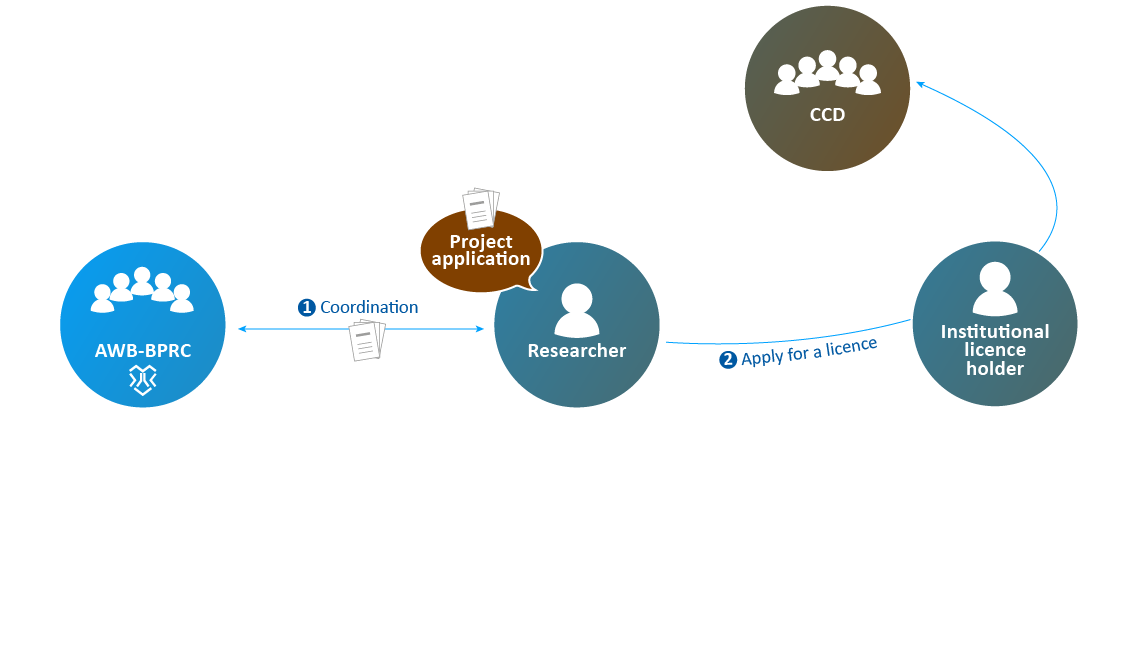
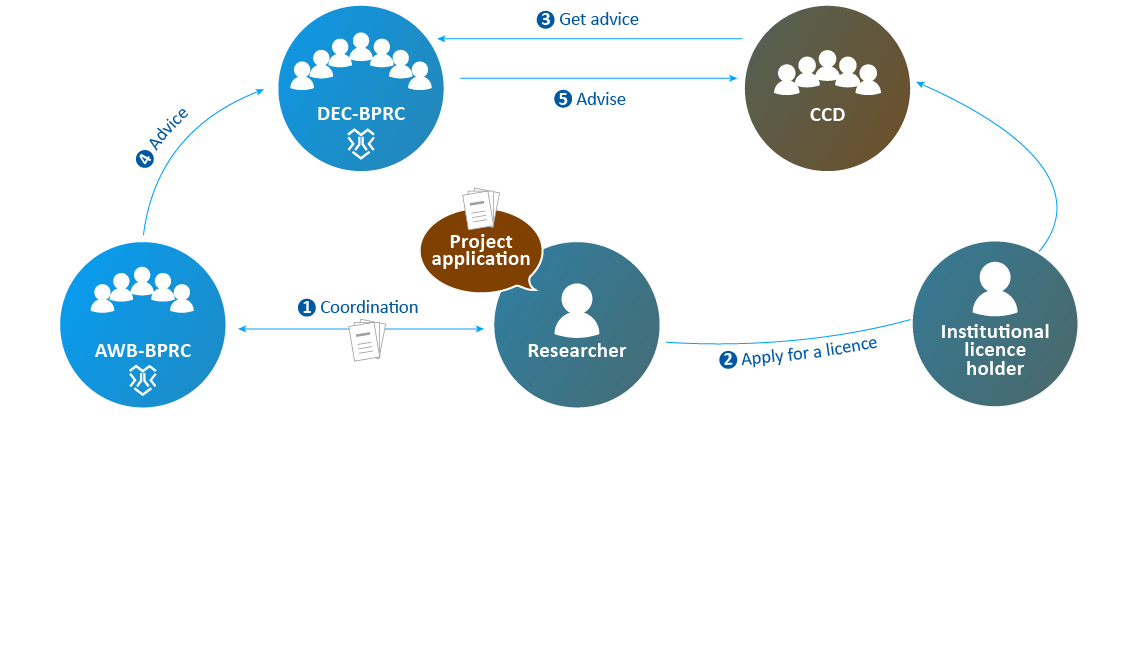
- The application is submitted to the AWB to assess clarity and technical feasibility.
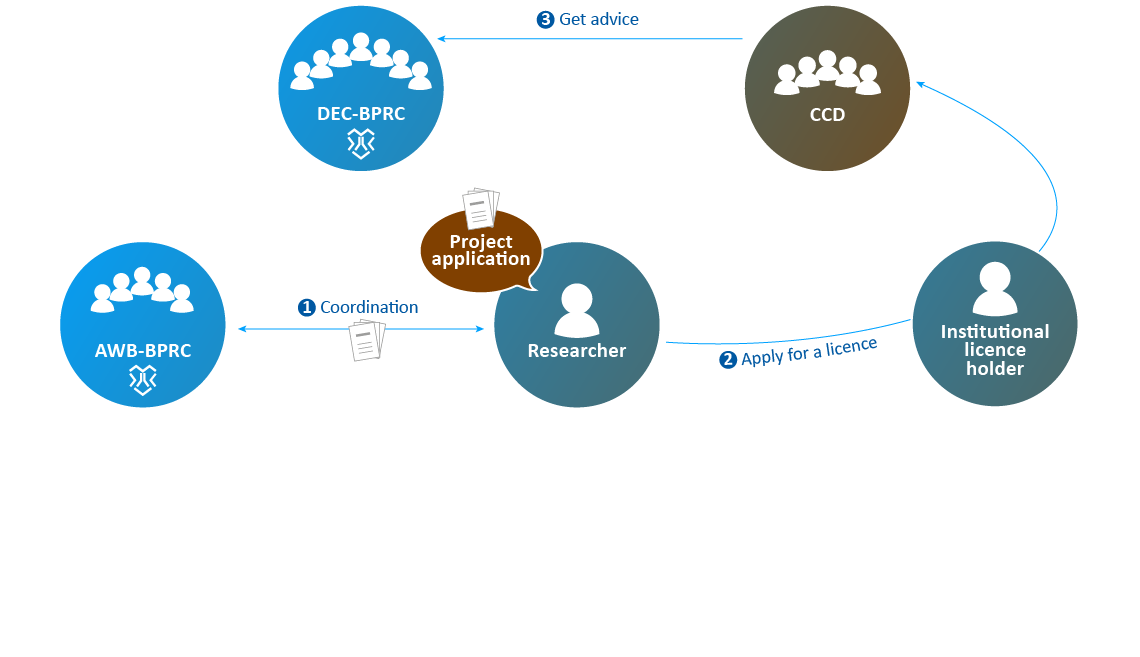
- After processing the feedback, the institutional licence holder submits the application to the CCD.
- The CCD selects a DEC and sends the application to that DEC to get advice. In 2019, the CCD submitted all applications filed by BPRC to the DEC-BPRC.
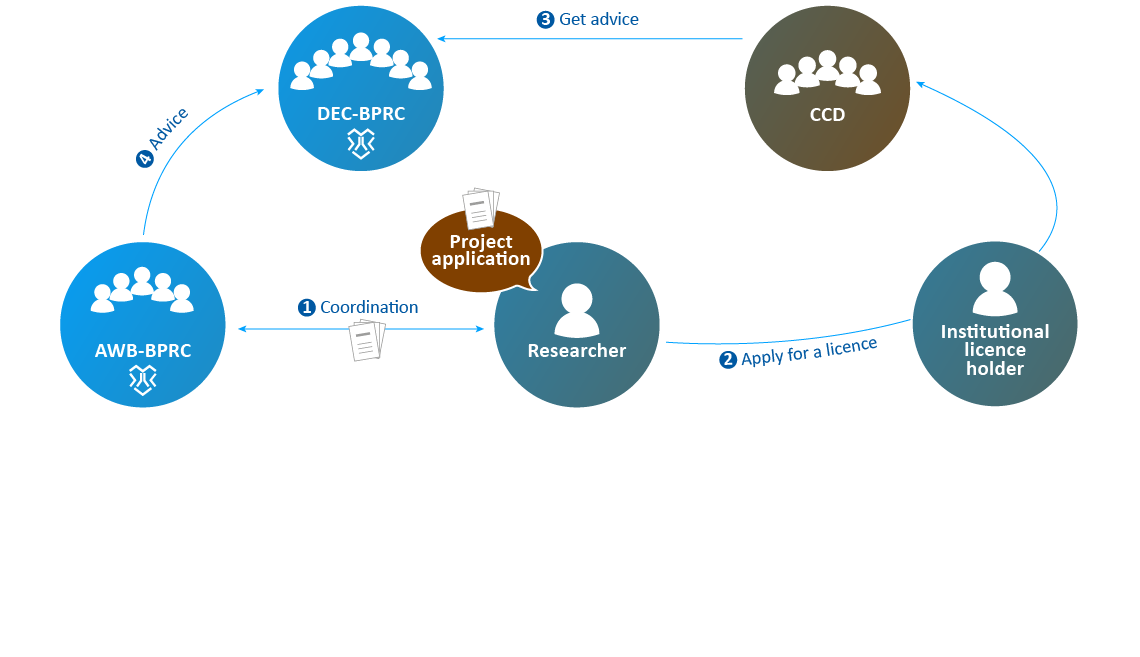
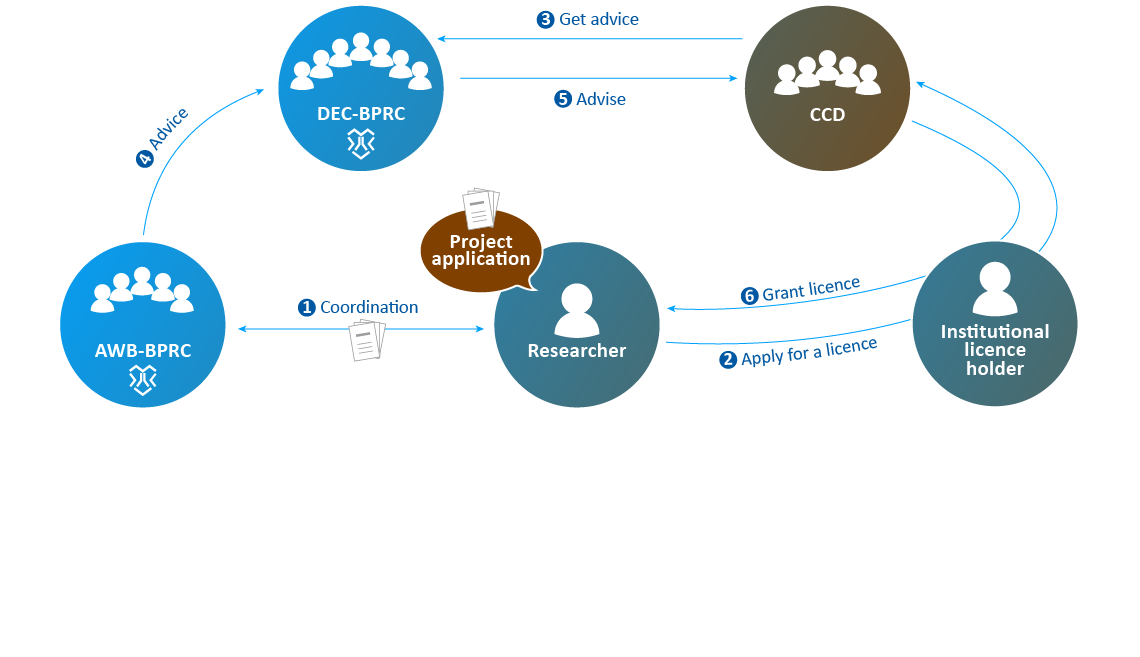
- The AWB advises the DEC if necessary.
- The DEC sends its advice to the CCD.
- The CCD decides on issuing a "project licence".
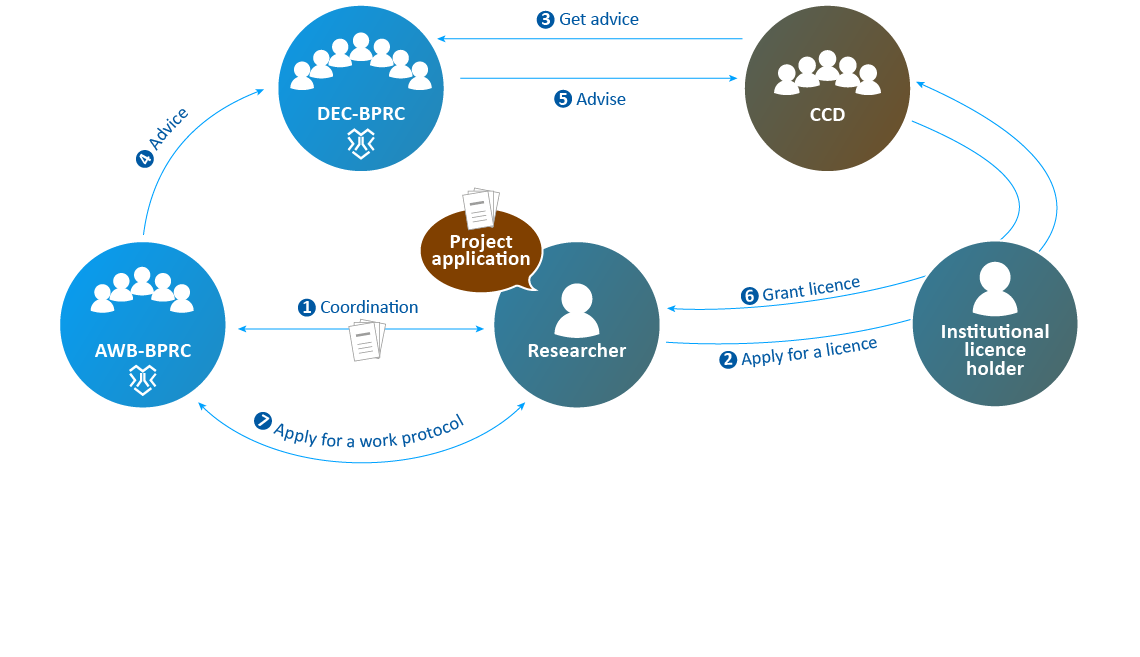
- After the licence is granted (AVD number), applications for individual animal experiments ("work protocol": CCD number) can be submitted to the AWB.
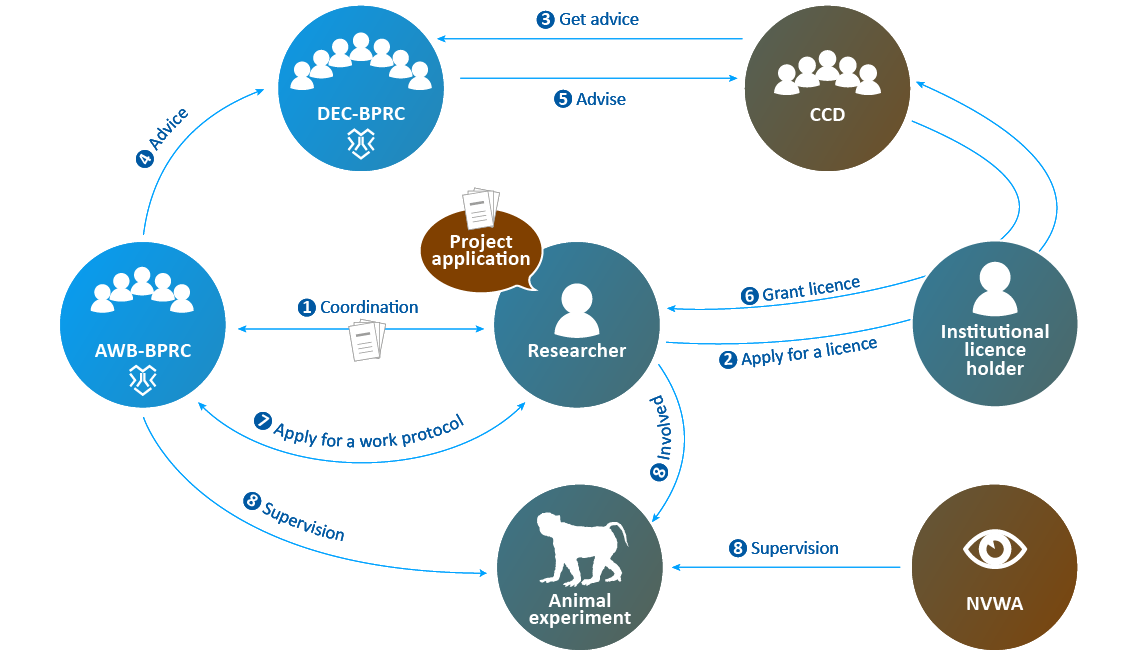
- When the AWB has evaluated whether the proposed study complies with the licence granted by the CCD, the applicant may conduct the experiment. The NVWA supervises the execution of the animal experiment.
-
Project licences
Project licences
Animal Ethics Committee (DEC) and Animal Welfare Body (AWB)
The Animal Ethics Committee (DEC) is responsible for ethically evaluating whether the implications of the proposed animal experiment for the health and welfare of the animals outweigh the scientific and social interests.
An important part of this ethical evaluation is to thoroughly explore whether alternative methods can be used for the proposed animal experiment. If the intended answers can also be obtained without using laboratory animals (replacement), by using fewer laboratory animals (reduction) or with a different experimental design in which the laboratory animals experience less discomfort (refinement), the DEC will advise against the proposal. As a result, the proposed animal experiment will not be conducted.
On 18 December 2014, the amended Dutch Experiments on Animals Act (WoD) came into force. Because of this act, the process to obtain permission for conducting an animal experiment has changed considerably. New authorities have been created, such as the Central Authority for Scientific Procedures on Animals (CCD) at a national level and the Animal Welfare Body (AWB) at an institutional level.
The AWB is an important link between the CCD and the researcher. Currently, the AWB at BPRC consists of the breeding colony manager (expert in animal behaviour), two scientists, a veterinarian and a biotechnician, all of them work at BPRC. In addition, several advisors are involved: a veterinarian with expertise in health care for laboratory animals, who also works at BPRC, and an animal welfare officer not affiliated with BPRC.
The mission of the AWB-BPRC is to aim for the best possible welfare of our laboratory animals in every aspect. This is done based on the principles of the 3Rs (Replacement, Reduction and Refinement), the intrinsic value of the animals and the current legal provisions. Furthermore, the AWB-BPRC supervises that the animal experiments are executed correctly within our institute.
The WoD dictates that an experiment involving laboratory animals may only be conducted after the CCD has issued a "project licence" to the license holder. The procedure is illustrated here.
The number of applications for project licences and work protocols can be found in our Annual report on laboratory animal science (Dutch), below the section Project licences.
Would you like to know more about the rules we comply with?
BPRC acts according to the Dutch Experiments on Animals Act (WoD). You can read more about this on the page Legislation and ethics.
Previous licences
Licences that have been granted to us are listed on Permits for animal studies.